  A weather indicator can be made with a hydrate of cobalt (II) chloride, which changes color as a result of the following reaction: Cl2 (s) Cl2 (s) +2H20(g)ĭoes a pink color indicate “moist” or “dry” air? Explain. What would be the effect on the equilibrium position of an equilibrium mixture of carbon, oxygen, and carbon monoxide if the total pressure of the system were decreased? 2C2 (s) +O2 (g) 2CO(g) RIGHT 10. What would be the effect on the equilibrium position of an equilibrium mixture of Br2, F2, and BrF5 if the total pressure of the system were decreased? 2BrF5(g) Br2(g) + 5F2 (g) RIGHT 9. In which direction would the equilibrium shift for the equation in 1f if heat were added? (the rxn is endothermic). In which direction would the equilibrium shift for the equation in 1e if CH3OH were removed? RIGHT 7. In which direction would the equilibrium shift for the equation in 1d if pressure were decreased? LEFT 6. In which direction would the equilibrium shift for the equation in 1c if O2 were removed? LEFT 5. In which direction would the equilibrium shift for the equation in 1b if N2O5 were added? RIGHT 4. In which direction would the equilibrium shift for the equation in 1a if S2 were removed from the reaction vessel? RIGHT 3. #Cobalt chloride weather indicator skinMAY CAUSE ALLERGIC SKIN OR RESPIRATORY REACTION.Equilibrium Problem Set #2 For each of the following reactions, describe what effect and increase in pressure would have on the equilibrium position of the reaction (i.e. CAUSES IRRITATION TO SKIN, EYES AND RESPIRATORY TRACT. WARNING! HARMFUL IF SWALLOWED OR INHALED. 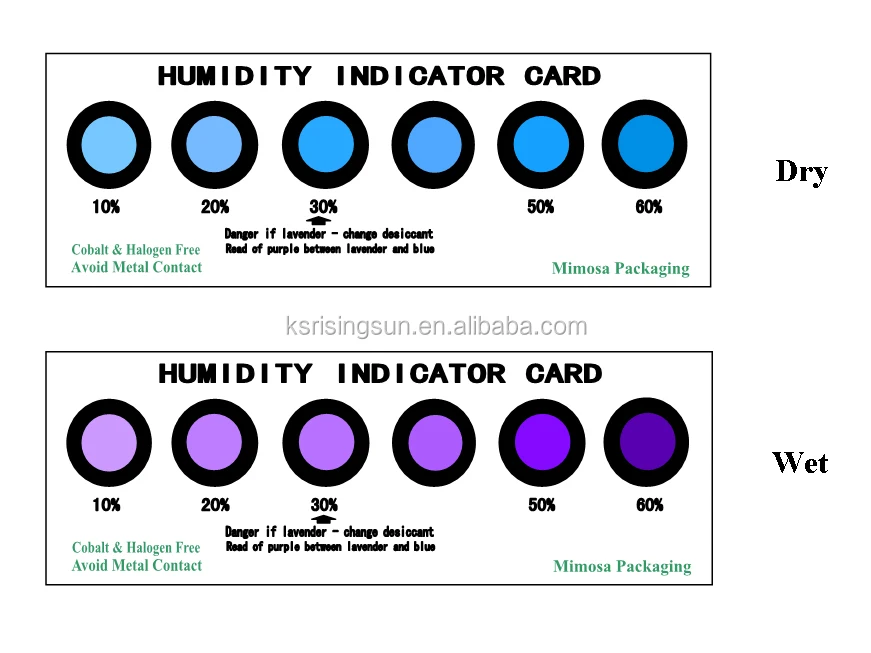 
The molecule has a Cobalt atom at its centre - Cobalt is a trace element so giving dietry supplements may be important. * Vitamin B12 is synthesized in the guts of animals by bacteria, it is not absorbed from food. Cobalt chloride can also be used in the magnetic tape industry where it can be used as a dopant.It is used in the textile industry where it acts as a mordant binding the colorant to the fiber.In the tire industry - it improves the bond between the steel and the rubber in steel radial tires.Food additive - a source of vitamin B12 *.Lab Protective Equip: GOGGLES LAB COAT VENT HOOD PROPER GLOVES Hmm, I haven't seen one of those for years - I wonder why.?Ĭontact Rating: 1 - Slight. Unfortunately humidity is not a very good predictor of the weather and I was soon disappointed with my new ' toy', although I still loved to see the colours change. Anhydrous cobalt chloride, CoCl 2 is a blue salt, but the the hydrated form, CoCl 2.6H 2O is red and a thin film of chemical on an ornament readily changes state depending on the humidity of the atmosphere. The magic, I found out some years later, was cobalt chloride, (or to be more specific, cobalt II chloride or cobalt dichloride). Blue was supposed to indicate good weather, pink was for rain. It was made of a chalky-white material and seemed to be brushed with magic! My beautiful ornament could change colour from pink, through various shades of lavender, to blue. Cobalt chloride first made an impact on my life when I was a small child - I was given a cheap little ornament that was supposed to be able to predict the weather. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
